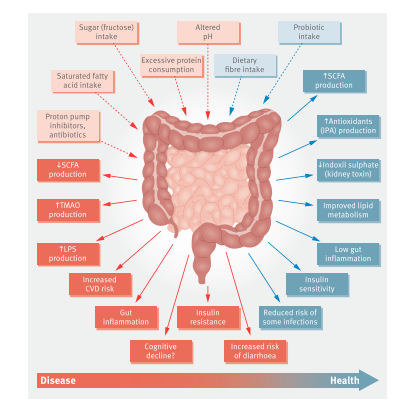
The gastrointestinal microbiota has been implicated in modulating human well-being and metabolism and is associated with chronic nutrition-related diseases such as obesity and diabetes. It has also been found to affect systemic functions such as immunity and cognitive abilities. The gut microbiota can impact health via the development of metabolites, such as short-chain fatty acids (SCFAs), that can affect a wide variety of host systems and metabolic pathways. The gut microbiota reacts to environmental stimuli rather than being a fixed characteristic. The effects of lifestyle factors, such as diet and exercise, on the gut microbiota have been the focus of much microbiome research. Variation in the gut microbiome's makeup and function has driven study on the relationship between aspects of the gut microbiota, such as diversity or the existence, absence, or quantity of certain taxa, and host well-being. In order to personalize dietary recommendations and avoid diet-related chronic diseases, precision nutrition research is also exploring how to anticipate individual variations in glycemic response, triglycerides, cholesterol levels, and other health indicators (Hughes, 2020).
In humans, the gut microbiota develops across the life span and plays a vital role in health and disease. The gut microbiota has various beneficial functions in wellness, including energy recovery from the metabolism of non-digestible food, defense against pathogens, and immune system modulation. A dysbiotic gut microbiota has a role in pathological disorders, such as obesity, diabetes, and atopy, irritable bowel syndrome, and inflammatory bowel disease (Jovel et al., 2018).
While long recognized for their value, research has generally overlooked the bacteria that comprise the gut microbiota. Scientists are now starting to work together to create personalized medicine microbiome research and perhaps biotherapeutics. Existing evidence confirms the importance of probiotics, prebiotics, and synbiotics in a wide variety of gastrointestinal disorders. Regarding the modulation of immune functions, there is evidence that probiotics can effectively prevent atopic eczema and help reduce sensitization. There is also evidence that enhancing the microbiota's colonization and succession in the first months and years of life can have effects beyond allergy prevention. These advantages may include the ability to decrease the susceptibility to respiratory tract infections, the likelihood of obesity, and the chance of acquiring autoimmune diseases. The potential for probiotics to provide benefits in health improvement and mitigation of disease risk in areas such as obesity, cardiovascular disease, and brain and neurological function supports the idea that the microbiota has a profound impact on human physiology. Probiotics and prebiotics can provide therapies to manipulate the microbiome, possibly opening up new health care treatment options (Bull & Plummer, 2015).
The fact that microbes in symbiosis are capable of colonizing nearly any space in the human body implies that they may have specific outbalanced modes of adaptation, the disruption of which can lead to fatal outcomes or disease. With diagnostic tools for earlier detection and prevention of illness, this disruption could be identified. In health research, the future aim is the modulation of health and disease states. New microbial treatment options can include microbes containing medicines or foods that may treat diseases (Hadrich, 2018).
Study into the gastrointestinal microbiota is still an evolving frontier. A significant proportion of the available research is difficult to translate into measurably successful treatments. However, straightforward dietary approaches such as adding a variety of brightly-colored fruits and vegetables rotated regularly can also be incredibly effective at improving the microbiome and well-being. The ideal personalization for diet and lifestyle medicine is individualized prescriptions of unique prebiotic compounds and probiotic strains; however, these are still somewhat futuristic (Toribio-Mateas, 2018).
References
- Bull, M. J., & Plummer, N. T. (2015). Part 2: Treatments for chronic gastrointestinal disease and gut dysbiosis. Integrative Medicine (Boulder), 14(1), 25–33.
- Hadrich, D. (2018). Microbiome research is becoming the key to better understanding health and nutrition. Frontiers in Genetics, 9(JUN), 1–10. https://doi.org/10.3389/fgene.2018.00212
- Hughes, R. L. (2020). A Review of the Role of the Gut Microbiome in Personalized Sports Nutrition. Frontiers in Nutrition, 6(January). https://doi.org/10.3389/fnut.2019.00191
- Jovel, J., Dieleman, L. A., Kao, D., Mason, A. L., & Wine, E. (2018). The Human Gut Microbiome in Health and Disease. Metagenomics: Perspectives, Methods, and Applications, 197–213. https://doi.org/10.1016/B978-0-08- 102268-9.00010-0
- Toribio-Mateas, M. (2018). Harnessing the Power of Microbiome Assessment Tools as Part of Neuroprotective Nutrition and Lifestyle Medicine Interventions. Microorganisms, 6(2), 35. https://doi.org/10.3390/microorganisms6020035
*These statements are not meant to diagnose or treat. You should consult your health care provider before starting any new diet, exercise, or supplement.